In 1998, a professor named Phil Allen snowshoed a plant science class into Rock Canyon above Provo at night. Headlamps on, we dug through the snowpack and into the frozen soil beneath a stand of Gambel oak, not because it was convenient, but because he thought the dark made it more memorable. He was right. When we scraped the dirt from those roots and held them under our headlamps, we were looking at something most people never see: pale fungal threads wrapped around each root tip and lacing through the soil between trees. We warmed up afterward with hot cocoa at the cars, but the image stuck. There is an entire economy happening underground, and most tree management never accounts for it.
Understanding this economy changes how you read a landscape. It explains why trees planted in stripped construction soil struggle for a decade. It explains why heavy fertilization can leave trees less drought-tolerant, not more. And it makes a strong case that the most useful thing you can do for the trees and shrubs in your care is not to add something. Stop doing several things you are probably already doing.
What Is Going On Underground
Every tree and shrub root system lives in partnership with soil fungi. The plant sends sugars downward, roughly 10 to 20 percent of everything it makes through photosynthesis goes to feed its fungal partners. The fungi, in return, deliver phosphorus, water, and other nutrients back up. It is a trade, not a parasite relationship: both organisms get something they could not access as efficiently on their own.
The practical benefit for the plant is reach. A root tip can only grow so far, and the finest root hairs (the actual absorbing surfaces) extend just a fraction of a millimeter into the surrounding soil. The fungal threads attached to those roots spread centimeters to meters outward, threading through pore spaces too small for any root to enter; this portion of the fungal network extending beyond the root is called the extramatrical mycelium. In effect, the fungi are the plant’s extended root system, accessing soil volume the roots themselves never touch.
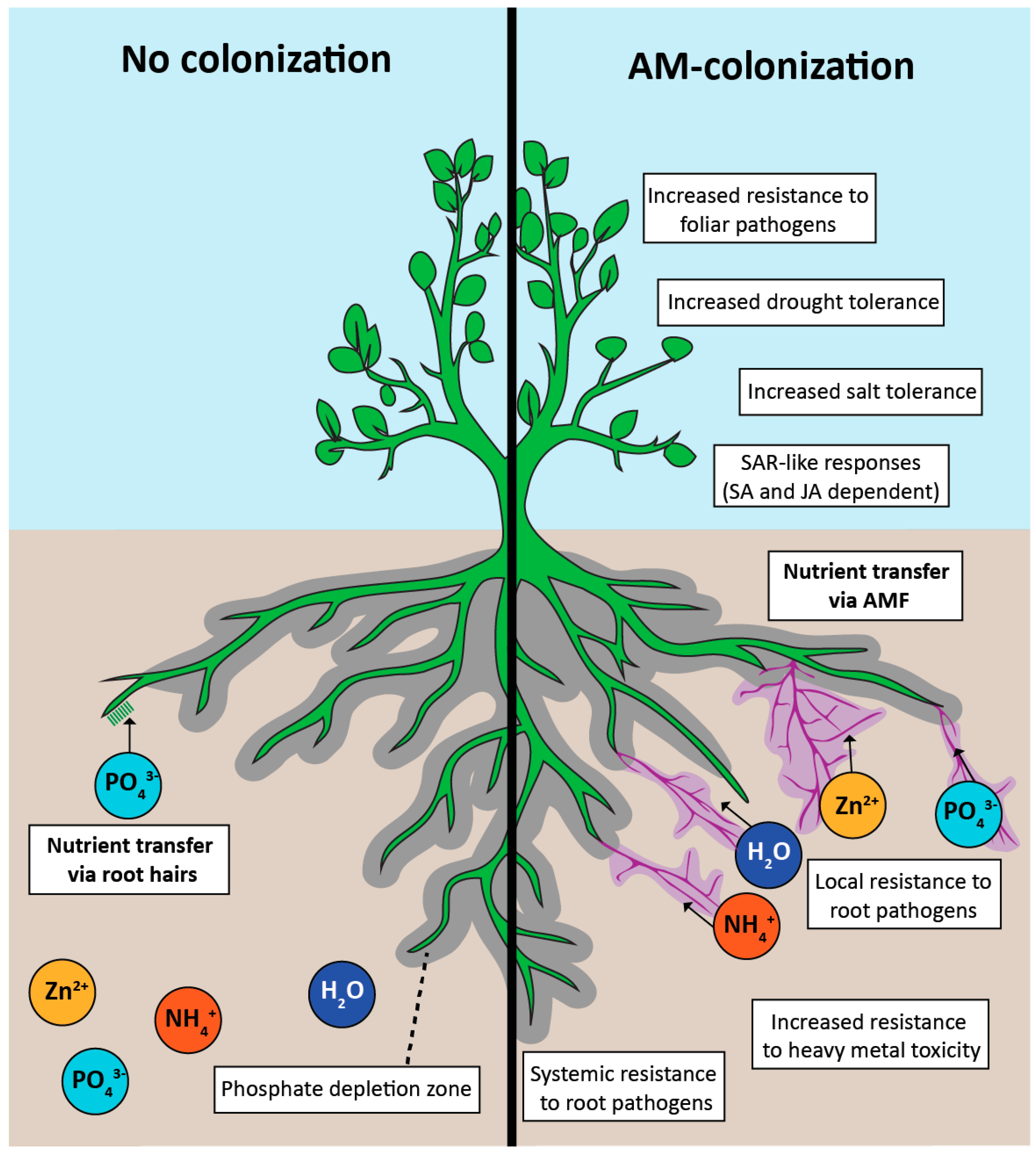 Left: a root without a fungal partner, limited to the small zone immediately around it. Right: the same root colonized by mycorrhizal fungi; the fungal threads extend far beyond that zone, pulling in phosphorus, zinc, and water from soil the root alone could never reach. Diagram: Jacott, Murray & Ridout, Agronomy 2017, CC BY-SA 4.0.
Left: a root without a fungal partner, limited to the small zone immediately around it. Right: the same root colonized by mycorrhizal fungi; the fungal threads extend far beyond that zone, pulling in phosphorus, zinc, and water from soil the root alone could never reach. Diagram: Jacott, Murray & Ridout, Agronomy 2017, CC BY-SA 4.0.
This matters most for phosphorus, which moves slowly through soil and gets depleted quickly near roots. It matters for water during late-summer drought. And it matters for trace nutrients that sit in the soil but require that extra reach to capture.
Two Types, and Which Plants Have Which
Not all mycorrhizal fungi work the same way. There are two main types, and knowing which your plants have tells you a lot about how to care for them.
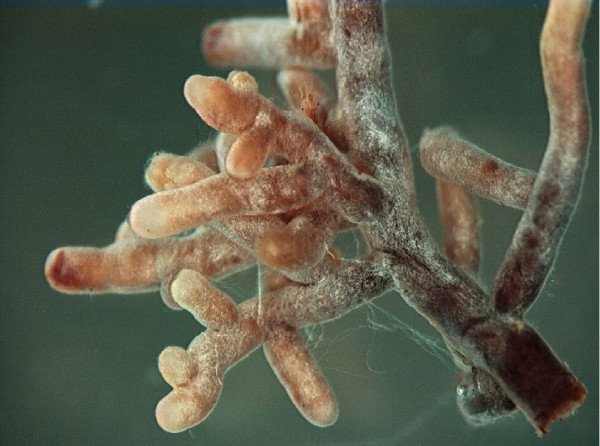
 Four forms of ectomycorrhizal root tips photographed under magnification; the white-sheathed form (lower left) is what you see when you dig into undisturbed conifer soil in the Puget Sound lowlands. Photo: Marc-André Selosse, CC0.
Four forms of ectomycorrhizal root tips photographed under magnification; the white-sheathed form (lower left) is what you see when you dig into undisturbed conifer soil in the Puget Sound lowlands. Photo: Marc-André Selosse, CC0.
The first type wraps around the root tip. These are called ectomycorrhizal fungi (ECM for short), and they coat each root tip in a visible pale sheath, exactly what we were looking at in Rock Canyon. The threads slip between the outer root cells but never enter them, forming a structure called the Hartig net where most of the nutrient exchange occurs. ECM fungi partner with conifers, oaks, birch, and a handful of other tree families. In the Puget Sound lowlands, that means Douglas-fir, western hemlock, Sitka spruce, western red cedar, all native and ornamental oaks, birch, alder, pine, hawthorn, hazel, larch, linden, walnut, and redbud. These are also the fungi responsible for most of our edible forest mushrooms: chanterelles, porcini, and Amanita are all ECM fungi fruiting from roots a few inches underground.
The second type goes inside the root cells. These are arbuscular mycorrhizal fungi (AM fungi), and they push their threads directly through the root cell wall, forming a highly branched structure inside each cell (called an arbuscule) where the actual nutrient exchange happens. This type is far more common: roughly 70 percent of all plant species rely on AM fungi, including maples, fruit trees, most ornamental shrubs and perennials, lawn grasses, and vegetable crops.
A third, less well-known group partners exclusively with plants in the heather family: salal, kinnikinnick, rhododendron, blueberry, madrone, and heather all depend on a specialized type called ericoid mycorrhizae. These plants evolved in acidic, high-organic soils with specific fungal communities. Strip that topsoil in construction grading and the fungi disappear with it; then wonder why the salal you replanted never took hold.
One more thing worth knowing: some plants form no mycorrhizal partnerships at all. Mustards, plants in the pink family, and plants in the goosefoot family fall into this category, and so do many of the weeds that move into disturbed ground. On a site that has been graded or heavily disturbed, these non-partnering weeds take over, and because their roots don’t support the fungi, the spore population in the soil gradually declines. When you later try to plant oaks or native conifers, the fungal partners those plants need may no longer be there in adequate numbers. This is one reason why restoration plantings on disturbed urban sites so often underperform: the soil biology was gone before the plants went in.
What the Fungi Do Beyond Feeding the Tree
Nutrient uptake gets most of the attention, but the fungi are doing several other things at the same time.
The physical mass of fungal threads in the soil helps hold it together. The threads produce sticky compounds (glomalin chief among them) that bind soil particles into clumps, improving drainage and making the soil less prone to compaction. A forest soil with a healthy fungal community is more resilient to disturbance than the same soil after that community is disrupted.
The sheath around ECM root tips also provides a physical barrier against some soil-borne pathogens. It is not immunity, but it is a layer of protection that disappears when fungal colonization drops.
You have probably also heard the “wood wide web” idea: that trees in a forest are connected through shared fungal networks, passing carbon and nutrients between them. The underlying science is real: researchers at the University of British Columbia traced carbon moving between Douglas-fir and birch trees through shared fungal threads in the early 1990s, and work done right here in the Pacific Northwest has confirmed that more than 95 percent of western hemlock root tips share fungal partners with surrounding Douglas-fir. The network infrastructure genuinely exists.
What the popular press has done with that finding is a different matter. The “mother tree nursing her seedlings” story has run ahead of the evidence. The amounts of carbon transferred between trees through these networks appear to be modest, real and measurable, but small relative to what a tree makes on its own. And several of the headline studies have been questioned on methodology. The science supports tree-to-tree fungal connections as ecologically meaningful, particularly in forest succession. It does not support the idea that forests operate as cooperative societies making conscious decisions to share resources. The reality is remarkable enough without the fable.
What Kills the Partnership
This is where the practical management implications start. Several common landscape practices damage mycorrhizal fungi, usually without any visible warning until the trees begin declining years later.
High-phosphorus fertilizer. When there is plenty of phosphorus already in the soil, a plant stops investing in its fungal partners; it no longer needs the fungi to find phosphorus, so it stops sending sugars down to feed them. The higher the available phosphorus, the weaker the fungal relationship becomes. Routinely fertilizing established trees with high-phosphorus formulations quietly undermines the fungal community over time, with no outward sign that anything is wrong until the tree is in trouble. A soil test before fertilizing is the only way to know whether you are correcting a real shortage or making one you cannot see.
Compaction. Fungi need oxygen in the soil to survive, just like roots do. When soil is compacted (by construction equipment, repeated foot traffic, or heavy vehicles) the air spaces collapse. Research in New Phytologist found that mycorrhizal function drops significantly at a bulk density of 1.60 Mg/m³ and disappears almost entirely at 1.75, thresholds that construction equipment reaches easily, and that heavy foot traffic on clay soils can approach over time. Once compaction reaches the point where root penetration stops, fungal growth stops too. The damage from a construction project compounds: roots are severed, the compacted soil prevents them from regenerating, and the fungal network that would have supported recovery is eliminated at the same time. For more on how roots respond to compaction, see How Tree Roots Grow and Develop.
Fungicides. Broad-spectrum fungicides kill fungi: pathogenic ones and beneficial ones alike. Applications timed for a leaf disease can suppress the mycorrhizal fungi in the root zone at the same time. This is a particular risk with preventive calendar-based spraying rather than targeted treatment of a diagnosed problem.
Topsoil loss. When soil is stripped by grading, rototilling, or excavation, the fungal threads are physically destroyed and the layer where fungal spores are most concentrated is removed. The subsoil underneath may have almost none. Recovery on a stripped site can take years even under the best conditions.
 Garlic mustard at full density in a deciduous forest understory. At this scale of invasion, the fungal community that native hardwood seedlings depend on for establishment is actively disrupted. Photo: Yehor Yatsiuk, CC0.
Garlic mustard at full density in a deciduous forest understory. At this scale of invasion, the fungal community that native hardwood seedlings depend on for establishment is actively disrupted. Photo: Yehor Yatsiuk, CC0.
Garlic mustard. This one is specific to our region. Garlic mustard (Alliaria petiolata) releases a chemical from its roots that disrupts the mycorrhizal fungi in the surrounding soil. Research from Harvard Forest has documented that heavy garlic mustard invasion effectively prevents the next generation of native canopy trees from getting established; the seedlings can germinate, but without a functioning fungal community they cannot thrive. Garlic mustard is spreading in Puget Sound lowland forest edges. The harm is not just competition for light; it is an attack on the soil ecosystem the next generation of oaks and maples depends on. Control efforts in forested areas are well worth the effort.
The Inoculant Question
You have probably seen mycorrhizal inoculant at the garden center, a powder or granule you mix in at planting, marketed as essential for healthy root development. For about a decade starting in the late 1990s, adding this product at planting was standard practice in landscape and nursery work. The recommendation made sense on paper: if mycorrhizal fungi are good for trees, adding more should help.
The research eventually caught up, and the results are not what the packaging implies.
The first problem is that established landscape soils already have fungal communities. When you introduce a commercial fungal strain into soil that already has native fungi living in it, the introduced strain has to compete against organisms that are already adapted to that exact location, that exact soil chemistry. The native fungi almost always win. A 2003 study in the ISA’s urban forestry journal applied inoculant to established street trees (pin oak, red maple, willow oak) and found no measurable growth benefit after a year.
The second problem is product quality. Researchers at the University of Kansas tested 23 commercial mycorrhizal inoculants and found serious problems: some products contained far fewer viable fungal spores than their labels claimed, some contained no viable spores at all, and a few were contaminated with a fungal pathogen that actually harmed the plants. A 2025 review of 302 product trials found that only 12 percent of commercial inoculants showed both a growth benefit and confirmed root colonization. Laboratory-produced inoculants used in research settings succeeded 63 percent of the time. The commercial market for these products is close to $1 billion a year globally; researchers estimated that roughly $876 million of that is spent on products that do not work as labeled. Unlike pesticides, mycorrhizal inoculants in the United States are not regulated for quality or efficacy.
The professional landscape industry has quietly stepped back from routine inoculant use as this evidence has built up. Consumer products are still widely sold.
There is a real use case, just a narrow one: sites where the native fungal community has been genuinely wiped out: severely graded construction subsoil, fumigated nursery ground, or mine reclamation sites. In those conditions, where the spore bank is gone and the soil has been essentially reset, inoculant can help. Even then, the best approach is low-tech: dig up a small amount of soil from beneath a healthy tree of the same species in a natural or undisturbed setting and mix it in at the planting hole. That soil carries locally adapted fungi without the quality control problems of commercial products.
How to Protect What Is Already There
The most effective mycorrhizal management strategy is not a product. It is a list of things to stop doing.
Mulch with arborist wood chips. A 4-inch layer of coarse, uncomposted wood chips (not bark nuggets, not sawdust) feeds the fungal community directly. The fungi colonize the woody material, break it down, and transfer nutrients to tree roots. This single practice does more for underground soil biology than almost anything else you can do in a managed landscape. It also moderates soil temperature, holds moisture, and suppresses weeds.
Fertilize only when a soil test says you need to. If there is already adequate phosphorus in your soil, adding more weakens the fungal partnership. When fertilization is genuinely warranted, choose slow-release or organic forms and avoid high-soluble-phosphate products around established trees and shrubs.
Use fungicides only for diagnosed problems. Preventive calendar spraying is the pattern most likely to harm mycorrhizal fungi as collateral damage. If a fungicide is necessary, apply it in response to a specific, identified pathogen, at the minimum rate that addresses the problem.
Protect the root zone from compaction. The rule of thumb for critical root zone (one foot of radius for every inch of trunk diameter) marks the minimum, not the full extent of the fungal influence, which extends well past the canopy edge. Before any construction or equipment work near trees, establish protection: fencing, wood chip surface protection, or both. Once the soil is compacted, restoring the fungal community takes years.
Leave the soil alone. Avoid rototilling around established trees and shrubs. When you need to replant a spot, go back into the existing soil rather than replacing it. The biology in undisturbed soil has been building for years; every time you disrupt it, you reset the clock.
The fungal community under your landscape has been developing since the first roots colonized that soil. Every year without a compaction event, without a heavy fertilizer application, without topsoil removal, is a year it gets stronger. The goal is not to introduce something new. It is to stop interrupting what is already working.
Sources:
- Mycorrhiza. Pacific Northwest Plant Disease Management Handbooks. Oregon State University / Washington State University.
- A Gardener’s Primer to Mycorrhizae (FS269E). Chalker-Scott, L. WSU Extension, 2017.
- Appleton, B. L., Koci, J., French, S., Lestyan, M., & Harris, R. (2003). Mycorrhizal fungal inoculation of established street trees. Arboriculture & Urban Forestry, 29(2), 107–111.
- Dawson, J. O. (2013). Things arborists should know about soil microbes. ISA Arborist News, February 2013, pp. 14–20.
- Koziol, L., Lubin, T., & Bever, J. D. (2024). An assessment of twenty-three mycorrhizal inoculants reveals limited viability of AM fungi, pathogen contamination, and negative microbial effect on crop growth for commercial products. Applied Soil Ecology, 202.
- Koziol, L., McKenna, T. P., & Bever, J. D. (2025). Meta-analysis reveals globally sourced commercial mycorrhizal inoculants fall short. New Phytologist.
- Nadian, H., Smith, S. E., Alston, A. M., & Murray, R. S. (1997). Effects of soil compaction on plant growth, phosphorus uptake and morphological characteristics of vesicular-arbuscular mycorrhizal colonization of Trifolium subterraneum. New Phytologist, 135(2), 303–311.
- Simard, S. W., Perry, D. A., Jones, M. D., Myrold, D. D., Durall, D. M., & Molina, R. (1997). Net transfer of carbon between ectomycorrhizal tree species in the field. Nature, 388, 579–582.
- Twieg, B. D., Durall, D. M., & Simard, S. W. (2007). Ectomycorrhizal fungal succession in mixed temperate forests. New Phytologist, 176(2), 437–447.
- Klein, T., et al. (2023). Belowground carbon transfer across mycorrhizal networks among trees: Facts, not fantasy. Open Research Europe.
- Invasive plant disrupts forest fungi. Harvard Forest press release. (Research on Alliaria petiolata and arbuscular mycorrhizal communities.)
Always read pesticide labels before use. WSU Extension has additional guidance on fungicide selection and timing for ornamental plantings.