You walked past it on a July morning and noticed the leaves looked tired. Not yellow exactly, not spotted, but pale and bronze across the upper surface like the green had been bleached out from below. You turned a leaf and saw the underside dotted with tiny black tar-spots and a few pinhead-sized insects clinging near the midrib. You searched “rhododendron lace bug.” Every site told you what to do about Stephanitis rhododendri. They told you the wrong story.
The pest doing this damage in Puget Sound yards is not the species the name implies. It is Stephanitis pyrioides, the azalea lace bug. It arrived in Washington in 2008, attacks both rhododendrons and azaleas, and runs two full generations plus a partial third here every year. The classical “rhododendron lace bug,” S. rhododendri, exists in the region but is uncommon, has only one generation per year, and shows up later in summer on rhododendrons only. The mismatch matters because the timing windows, the host expectations, and the response are different for each species.
What You Are Looking At
 Stippled and bleached azalea foliage. The pale, washed-out leaves are the visible end of weeks of lace bug feeding from below. Photo: Janet Guynn, Alabama Cooperative Extension System (ACES), CC0.
Stippled and bleached azalea foliage. The pale, washed-out leaves are the visible end of weeks of lace bug feeding from below. Photo: Janet Guynn, Alabama Cooperative Extension System (ACES), CC0.
Look at the upper surface of an affected leaf. The damage is stippling, pale dots where lace bug mouthparts pierced through the leaf from below and drained out cell contents one cell at a time. Light infestations show as scattered pinprick speckling. Heavier feeding turns the whole upper surface silvery, yellowish, or bronze, and the leaf takes on a dull, washed-out look that contrasts sharply with healthy growth nearby. Damaged leaves never recover. They hang on through the season looking worse each week, then drop in fall earlier than normal foliage.
Turn the leaf over and the diagnosis confirms itself. The undersides carry dark, tar-like fecal deposits stuck to the surface in irregular spots, especially clustered along the midrib and major veins. Look closer with a hand lens and you will find the bugs themselves: nymphs are tiny, spiny, dark-colored, and clustered in groups along the leaf veins. Adults are about an eighth of an inch long, flat, with lacy transparent wings held flat over the back.
 The diagnostic underside view. Dark tar-spot fecal deposits along the veins confirm lace bug rather than mites or chlorosis. Photo: Janet Guynn, Alabama Cooperative Extension System (ACES), CC0.
The diagnostic underside view. Dark tar-spot fecal deposits along the veins confirm lace bug rather than mites or chlorosis. Photo: Janet Guynn, Alabama Cooperative Extension System (ACES), CC0.
 A heavily fed-on rhododendron leaf, Soos Creek Botanical Garden, late April 2026. The yellow color reads as chlorosis at a glance; the dark spotting is the diagnostic that tells you it is not. Photo: Chris Welch, Soos Creek Botanical Garden, Auburn, WA.
A heavily fed-on rhododendron leaf, Soos Creek Botanical Garden, late April 2026. The yellow color reads as chlorosis at a glance; the dark spotting is the diagnostic that tells you it is not. Photo: Chris Welch, Soos Creek Botanical Garden, Auburn, WA.
Two look-alikes send people down the wrong path.
Advanced lace bug damage mimics iron-deficiency chlorosis. Both turn rhododendron leaves yellow. True chlorosis leaves the underside clean and shows green veins against the yellowing lamina; it comes from soil pH too high for the plant to take up iron, not from feeding insects. Lace bug damage on the same plant, yellowing accumulated from weeks of cell-content drainage, looks similar at a glance, but the leaves carry the dark tar-spot deposits visible above. If your rhody is yellowing and the undersides are clean, the problem is the soil. See the soil pH amendment guide for that path. If the undersides look like the leaf above, it is lace bug, and the infestation is well past the easy intervention window.
Spider mites also produce stippling on rhododendron and azalea, but their damage is finer and more uniform, and there are no fecal deposits on the undersides. Mites sometimes leave fine webbing. If you see stippling without tar spots and without visible bugs, mites are the likely cause and the treatment is different.
The Three Species, and Why You Care
Three lace bug species turn up on broadleaf evergreens in the region. They look nearly identical at a glance and get conflated in casual descriptions, but they behave differently enough that treating them as one pest leads to wrong-timed sprays and missed windows.
 Adult and nymph on an azalea leaf, with a centimeter ruler showing the adult is roughly 3 mm long. The lacy reticulated wings and the brown thoracic markings identify Stephanitis pyrioides. Photo: Forest and Kim Starr, Makawao, Maui, CC BY 3.0 US.
Adult and nymph on an azalea leaf, with a centimeter ruler showing the adult is roughly 3 mm long. The lacy reticulated wings and the brown thoracic markings identify Stephanitis pyrioides. Photo: Forest and Kim Starr, Makawao, Maui, CC BY 3.0 US.
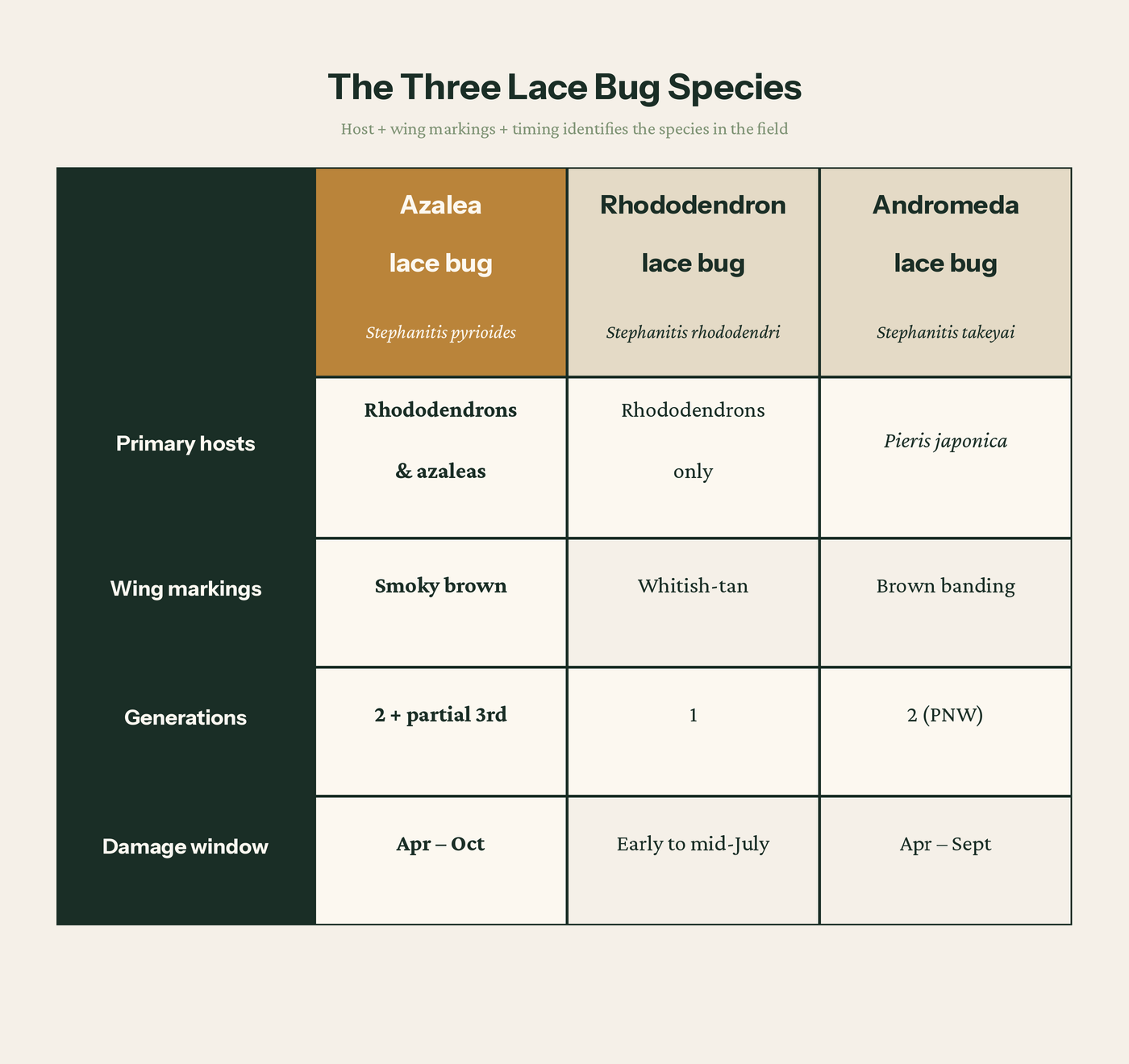 Three lace bug species turn up here. The azalea lace bug is the one doing almost all of the damage on rhododendrons and azaleas in Puget Sound.
Three lace bug species turn up here. The azalea lace bug is the one doing almost all of the damage on rhododendrons and azaleas in Puget Sound.
Stephanitis pyrioides, the azalea lace bug. Hosts: both rhododendrons and azaleas. Wing markings: smoky brown. Two full generations plus a partial third here every year. This is the species responsible for almost all “rhododendron lace bug” complaints in the region, and the strongest peer-reviewed lifecycle data for our climate comes from University of Washington work on this species. Damage starts in spring, intensifies through summer, and accumulates over repeated years to the point of plant decline on stressed shrubs.
Stephanitis rhododendri, the true rhododendron lace bug. Hosts: rhododendrons only. Wing markings: whitish-tan, no smoky patches. One generation per year, with damage typically visible early to mid-July. WSU HortSense is clear on this distinction: S. rhododendri does not feed on azaleas. It is much less commonly reported than S. pyrioides in the region. Treatment timing mirrors S. pyrioides but shifted later, target nymphs in late June and early July with the same contact options.
Stephanitis takeyai, the andromeda lace bug. Hosts: primarily Pieris japonica. Wing markings: brown banding. Emerges later than the azalea lace bug, around hawthorn first bloom. If you have stippled foliage on Pieris and clean rhododendrons next to it, the bug is almost certainly S. takeyai, not S. pyrioides.
Host plus wing markings plus timing identifies the species in the field.
What It Eats Here That the Literature Missed
The national guides stop at rhododendron and azalea. The PNW story is wider. Cage trials at Oregon State University and the Oregon Department of Agriculture documented S. pyrioides feeding and reproducing on a string of regional natives and crops that national resources never mention.
 Salal (Gaultheria shallon), a common understory native that Oregon cage trials confirmed as a host. Photo: Peter Pearsall, U.S. Fish and Wildlife Service, public domain.
Salal (Gaultheria shallon), a common understory native that Oregon cage trials confirmed as a host. Photo: Peter Pearsall, U.S. Fish and Wildlife Service, public domain.
Salal (Gaultheria shallon) and evergreen huckleberry (Vaccinium ovatum) supported full reproduction in cage trials. Both are major understory components of coastal and lowland forests in this region; they are also cut and sold as seasonal greenery. Highbush blueberry (Vaccinium corymbosum) and cranberry (V. macrocarpon) sustained feeding and reproduction, with damage observed on commercial plantings. Western azalea (Rhododendron occidentale), our only native rhododendron of the deciduous-azalea type, showed severe damage in ornamental plantings. Kalmiopsis species, vulnerable PNW endemics, were killed outright in trials.
Rhododendron lace bug in this region is not strictly a pest of ornamental rhodies. If you manage native plantings, restoration sites, or commercial small-fruit crops, watch for stippled foliage and tar-spot deposits across the broader Ericaceae list.
The Lifecycle, in Numbers
Each female S. pyrioides deposits up to 300 eggs over her adult life, inserting them one at a time into the underside of a leaf along the midrib and capping each with a varnish-like fecal shield that protects them through winter. Eggs hatch in spring as temperatures accumulate. Nymphs pass through five increasingly larger spiny instars before molting into adults. Total egg-to-adult development takes about 22 days at 86°F and 97 days at 59°F, and fails at 91.4°F or above. The lower temperature threshold for egg development is 50.4°F; for complete egg-to-adult development it is 52.2°F (Braman et al. 1992, the foundational thermal-requirements study for this species). Adults are non-diapausing, which means in our mild maritime winters some adults may persist longer than the older eastern-U.S. literature describes.
Published degree-day models use base temperatures near 50°F. Garrison & Tobin’s 2022 University of Washington work places 50 percent first-generation nymph emergence at 171 GDD base 50.4°F. The older mid-Atlantic figure for hatch completion is 214 GDD base 50°F (UMD IPMnet; Rosetta at OSU reports 213 GDD base 50.4°F). These models were built in climates where winter contributes little heat above their base threshold.
In the Puget Sound lowlands, base 50°F models are poor predictors of calendar date. Our mild maritime winters bank enormous heat between 32°F and 50°F that a base-50 model ignores entirely. By late March, Kent has accumulated roughly 800 GDD₃₂ but essentially zero GDD₅₀; a base-50 model would not start its clock for weeks. The published GDD₅₀ thresholds do not convert to GDD₃₂ by any stable ratio because the relationship depends on how cool days distribute across the season, and our cool-day profile is fundamentally different from the mid-Atlantic’s.
For regional timing we use GDD base 32°F benchmarks derived directly from six years of Puget Sound weather data (2021 through 2026) cross-referenced against field reports and indicator-plant phenology. Two benchmarks bracket the intervention window: 800 GDD₃₂ marks scouting onset (first nymphs reliably present), and 1,099 GDD₃₂ marks approximate hatch completion (last effective window for Gen 1 contact applications). See the growing degree days guide for the underlying method.
Puget Sound station data from 2021 through 2026:
| Year | Scouting onset (800 GDD₃₂) | End of hatch (1,099 GDD₃₂) |
|---|---|---|
| 2021 | March 26 | April 17 |
| 2022 | March 26 | April 19 |
| 2023 | March 31 | April 25 |
| 2024 | March 21 | April 10 |
| 2025 | March 29 | April 15 |
| 2026 | March 14 | April 3 |
Scouting onset in this region is reliably late March, and Gen 1 hatch is essentially complete by mid-April. The 2026 spring ran roughly two weeks ahead of normal across both columns. This is the regional anchor that no national lace bug guide carries: by the time mid-Atlantic publications start recommending May treatments, our first-generation nymphs are already adults. Check the current GDD₃₂ for your nearest station on the weather page to see where this season is tracking.
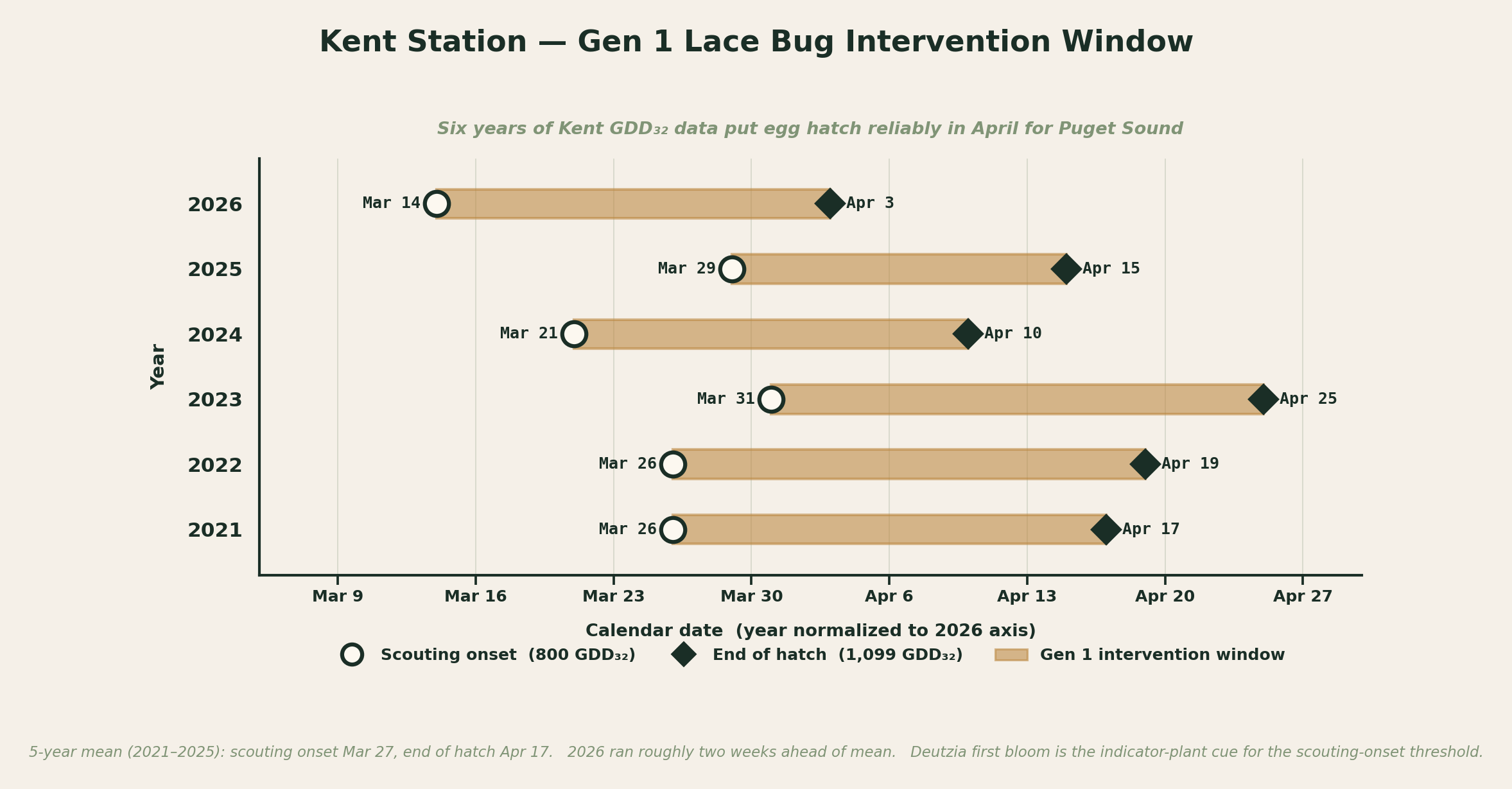 Six years of Kent GDD₃₂ data. Each tan band is that year’s Gen 1 intervention window, opening at the 800 GDD₃₂ scouting onset and closing at the 1,099 GDD₃₂ end-of-hatch threshold. 2026 ran roughly two weeks ahead of the 5-year mean.
Six years of Kent GDD₃₂ data. Each tan band is that year’s Gen 1 intervention window, opening at the 800 GDD₃₂ scouting onset and closing at the 1,099 GDD₃₂ end-of-hatch threshold. 2026 ran roughly two weeks ahead of the 5-year mean.
When to Scout, When to Treat
By the time upper-surface stippling is obvious, Gen 1 has been feeding for two to four weeks and Gen 2 is already forming. Scouting beats observation every year. Start inspecting leaf undersides weekly around Deutzia first bloom, which lines up with the 800 GDD₃₂ scouting onset, typically late March in average years and earlier in warm years like 2026. Check plants in sun first because lace bug populations build heaviest there. A hand lens helps, but tar-spot fecal deposits are visible to the naked eye and confirm the diagnosis even when the bugs themselves are hard to spot.
There is no published action threshold for ornamental plantings. The working version is qualitative: when nymphs are visible on multiple shoots, treat. Waiting for upper-leaf damage means treating Gen 2 and 3 with a population already established, which costs more product and more passes for less effect.
The late-April-through-May window is the one that matters. Hitting Gen 1 nymphs with a contact application before they molt to adults prevents the egg-laying that drives the summer generations. The summer-cleanup window in June through August is secondary, useful for keeping Gen 2 pressure off plants that escaped April treatment but never as efficient. Fall sanitation in September and October is third in order of importance and aimed at reducing overwintering pressure on plants you intend to keep.
Cultural Management
Plant placement is the most durable lever you have. Plants in full sun support heavier lace bug populations than plants in partial shade, and the mechanism is biocontrol, not plant stress. Research from UMass and UC IPM shows that natural enemy complexes, lacewings, predatory bugs, lady beetles, parasitic wasps, jumping spiders, are more abundant and more effective in shaded, structurally diverse landscapes. The standard advice “grow azaleas in shade” works because shade brings the predator community with it. Adding shade alone to a bare landscape does less than adding shade plus planting diversity.
Site rhododendrons and azaleas in partial shade where possible. Surround them with a mix of flowering shrubs and perennials rather than isolated specimens in lawn. Maintain steady soil moisture, drought stress amplifies lace bug damage. If you have a chronically damaged plant in full sun and the lace bug pressure returns every year, the durable fix is usually relocation or replacement, not annual treatment.
Resistant species exist. Among deciduous azaleas, Rhododendron atlanticum, R. arborescens, R. canescens, R. periclymenoides, and R. prunifolium show meaningful resistance in eastern trials. A 2009 USDA-ARS study confirmed lace-bug resistance in eleven Encore azalea cultivars, with Autumn Royalty and Autumn Twist among the most widely available in regional nurseries. Resistance work on evergreen rhododendrons is thinner and points to cultivar-by-cultivar variation rather than a clean resistant list, so site selection and predator-friendly landscape design carry more weight than chasing a specific cultivar. If you are replanting after losing a sun-exposed rhododendron to repeated lace bug pressure, consider mahonia, hydrangea, camellia, osmanthus, daphne, or kalmia as shade-tolerant alternatives.
Biological Management
Native predators in the region include green lacewings, assassin bugs, minute pirate bugs, lady beetles, jumping spiders, and predatory mites. None of them produce reliable single-handed control when populations are already high, but conserved predator communities suppress new infestations before they explode. Two of the specialist natural enemies effective against S. pyrioides in the eastern United States, the parasitoid Anagrus takeyanus and the predatory bug Stethoconus japonicus, are not established in the region based on current records (Rosetta, OSU NWREC). Plan around what is present, not what is theoretically available.
Augmentative release of green lacewing larvae is the only biocontrol option with quantitative backing for this pest. Research on nursery azaleas (Shrewsbury and Smith-Fiola, 2000) shows that releasing lacewing larvae at five per plant reduced lace bug populations by 79 percent, and ten per plant reduced them by 88 percent. The acephate comparison in the same trial gave 100 percent control. Later potted-azalea trials with Chrysoperla rufilabris in the same research line reported reductions up to 97 percent. Chemical is faster and cleaner. Biocontrol compounds year over year as predators establish. For high-value specimens, a well-timed lacewing release in April or May is a defensible choice; eggs are available from most beneficial-insect suppliers.
Chemical Management
Contact insecticides are the first chemical tier. Insecticidal soap, horticultural oil, neem oil, azadirachtin, and pyrethrin all kill nymphs on contact. Coverage of leaf undersides is essential because the bugs feed there. Multiple applications at 7 to 10 day intervals through the Gen 1 window controls the generation. Soaps may need three or more passes. Some azalea cultivars are soap-sensitive, test on a small section first.
Systemic neonicotinoid insecticides are the second tier and the more powerful option for severe infestations. Soil-drench applications of imidacloprid (Bayer Advanced Tree & Shrub, Merit) or dinotefuran (Safari) provide season-long control from one spring application before bloom. The constraint is pollinator safety: do not apply systemics to plants that will flower during the residual period, because the neonicotinoid translocates into nectar and pollen and persists for months to years in some plants. For rhododendrons and azaleas that bloom in spring, this rules out drench applications shortly before or during bloom on those same plants. The systemic option is most appropriate for established non-flowering specimens of secondary hosts.
Avoid foliar broad-spectrum sprays of carbamates, organophosphates, and synthetic pyrethroids. UC IPM is direct on this point: these products kill natural enemies, often trigger secondary spider mite outbreaks, and contaminate surface water. They are the most disruptive option for a pest that rewards conservation of predators.
Your Year, in One Page
| When | What | Why |
|---|---|---|
| November to February | Plan placement, list last year’s worst plants | Eggs are protected and unreachable, the work this season is decision-making |
| Late March, Deutzia bud break | Begin weekly scouting on plants in sun | Kent crosses the 800 GDD₃₂ scouting onset between mid-March and early April depending on the year |
| Mid-April, Deutzia first bloom | Treat Gen 1 nymphs with soap, oil, or neem | Kent crosses the 1,099 GDD₃₂ end-of-hatch around this date, last chance to hit Gen 1 before adults disperse |
| Late April to May | Repeat contact applications at 7-10 day intervals | Multiple applications are required, no contact spray gives one-shot control |
| April to May | Consider lacewing release on high-value specimens | Quantitative biocontrol option, compounds over years with conserved predator community |
| June to August | Continue scouting, suppress Gen 2 buildup as needed | Overlapping generations mean continuous nymph presence, contact treatments still effective |
| Late July | Watch for S. rhododendri damage on rhody-only plants | The true rhododendron lace bug, if present, becomes visible separately at this point |
| September to October | Prune damaged shoots, document affected plants | Females are setting next year’s overwintering eggs, sanitation reduces spring pressure |
Sources
- Garrison, R.R. and Tobin, P.C. 2022. Development of Azalea Lace Bug, Stephanitis pyrioides, on Susceptible and Resistant Rhododendron Species in Western Washington. Journal of Economic Entomology 115(1): 233–243, the peer-reviewed source for the two full plus partial third generation count in this region and the locally calibrated degree-day model.
- Braman, S.K., A.F. Pendley, B. Sparks, and W.G. Hudson. 1992. Thermal Requirements for Development, Population Trends, and Parasitism of Azalea Lace Bug. Journal of Economic Entomology 85(3): 870–877, the foundational study for development thresholds (50.4°F egg, complete development at 394 degree-days base 52.2°F) and the full lifecycle parameters that downstream extension publications draw from.
- Neal, J.W. and L.W. Douglass. 1988. Development, Oviposition Rate, Longevity, and Voltinism of Stephanitis pyrioides, an Adventive Pest of Azalea, at Three Temperatures. Environmental Entomology 17(5): 827–831, four-generation documentation for Maryland, non-diapausing adult finding, and oviposition rate data.
- Stephanitis pyrioides Scott, 1874 phenology in Oregon. 2019. Pan-Pacific Entomologist 95(2): 99, a second peer-reviewed PNW phenology data point that supports the regional generation count and timing observations.
- Eigenbrode, S.D., et al. 2020. Attracting Chrysopidae With Plant Volatiles for Lace Bug Control in Rhododendrons and Azaleas. Journal of Insect Science, quantitative biocontrol research using plant volatile attractants.
- WSU HortSense: Azalea: Azalea lace bug, regional fact sheet covering identification, lifecycle, and Washington-approved products.
- WSU HortSense: Rhododendron: Rhododendron lace bug, the regional reference for S. rhododendri identification and one-generation-per-year lifecycle.
- PNW Insect Management Handbook: Azalea and Rhododendron Lace Bug, the regional management handbook entry.
- OSU Solve Pest Problems: Azalea Lace Bugs, homeowner-facing OSU resource including iron deficiency look-alike comparison and resistant cultivar notes.
- Rosetta, R. Azalea Lace Bug IPM Presentation, OSU NWREC, the source for Washington 2008 and Oregon 2009 detection dates and novel host trials.
- Shrewsbury, P.M. and Smith-Fiola, D.C. 2000. Evaluation of green lacewings for suppressing azalea lace bug populations in nurseries. Cited in Rosetta presentation; source for the quantitative lacewing release reductions.
- UC IPM Pest Notes 7428: Lace Bugs, the standard non-PNW IPM treatment with the natural-enemy mechanism behind sun-versus-shade siting.
- UMass Extension: Stephanitis pyrioides, Mid-Atlantic comparison data including resistant deciduous azalea species and the landscape-complexity natural enemy finding.
- NC State Extension: Azalea Lace Bug, southeastern reference with the 300-egg fecundity figure and varnish-cap egg description.
- Wang, Y., C.D. Robacker, and S.K. Braman. 2009. Identification of Resistance to Azalea Lace Bug Among Deciduous Azalea Taxa. Journal of the American Society for Horticultural Science 134(1): 40–47. USDA-ARS study confirming lace-bug resistance in eleven Encore azalea cultivars.
