You get a soil test back and it says 5.6. You search the internet and every result tells you to lime. Raise the pH, they say. Get it closer to neutral. Your vegetables need 6.5. Your lawn needs 6.0. You should have been liming this whole time.
Stop. That advice was written for Ohio.
In the Puget Sound lowlands, your soil is already acidic because it is supposed to be. Decades of rainfall leaching and centuries of conifer litter decomposition have pushed native soil pH into the 5.0 to 6.0 range across most of the region. That is exactly where rhododendrons, blueberries, heathers, conifers, and most of the plants that define this landscape evolved to grow. A reading of 5.6 on Alderwood or Woodinville soil is not a problem to solve. It is the baseline.
The question is not “how do I fix my pH?” It is “does my pH need fixing at all?” Most of the time the answer is no. When it is yes, the fix depends on what you are growing, what soil you are standing on, and what pushed the pH out of the range your plants need.
Why the Number Matters
You have probably seen it on a rhododendron planted next to a house foundation: the leaves turn yellow between the veins while the veins themselves stay green. The plant looks sick. You fertilize it. Nothing changes. You fertilize it again. Still yellow. The iron the plant needs is six inches away in the soil. The roots cannot touch it.
That is a pH problem. The concrete foundation has been leaching calcium into the bed for years, pushing the soil pH above 7.0. At that pH, iron converts from a form roots can absorb into a form they cannot. The nutrient is there. The chemistry locks it away.
pH measures how acidic or alkaline your soil solution is, on a scale from 0 to 14. Seven is neutral. Below 7 is acidic. Above 7 is alkaline. The scale is logarithmic, which means each whole number is a tenfold difference: soil at pH 5.0 is ten times more acidic than soil at 6.0, and a hundred times more acidic than soil at 7.0. Small numbers on your test report represent large differences in soil chemistry.
What makes this matter for every plant in your yard is nutrient availability. Most essential nutrients are most accessible to roots between pH 6.0 and 7.0. Below 5.5, aluminum and manganese become soluble enough to reach toxic levels, stunting roots and killing fine root tips. Phosphorus locks up in forms roots cannot absorb. Above 7.0, iron, manganese, and zinc become increasingly unavailable, which is the mechanism behind those yellow rhododendron leaves. The iron chlorosis you see on rhododendrons, azaleas, pieris, and pin oaks near foundations is almost always a pH story, not a nutrient story.
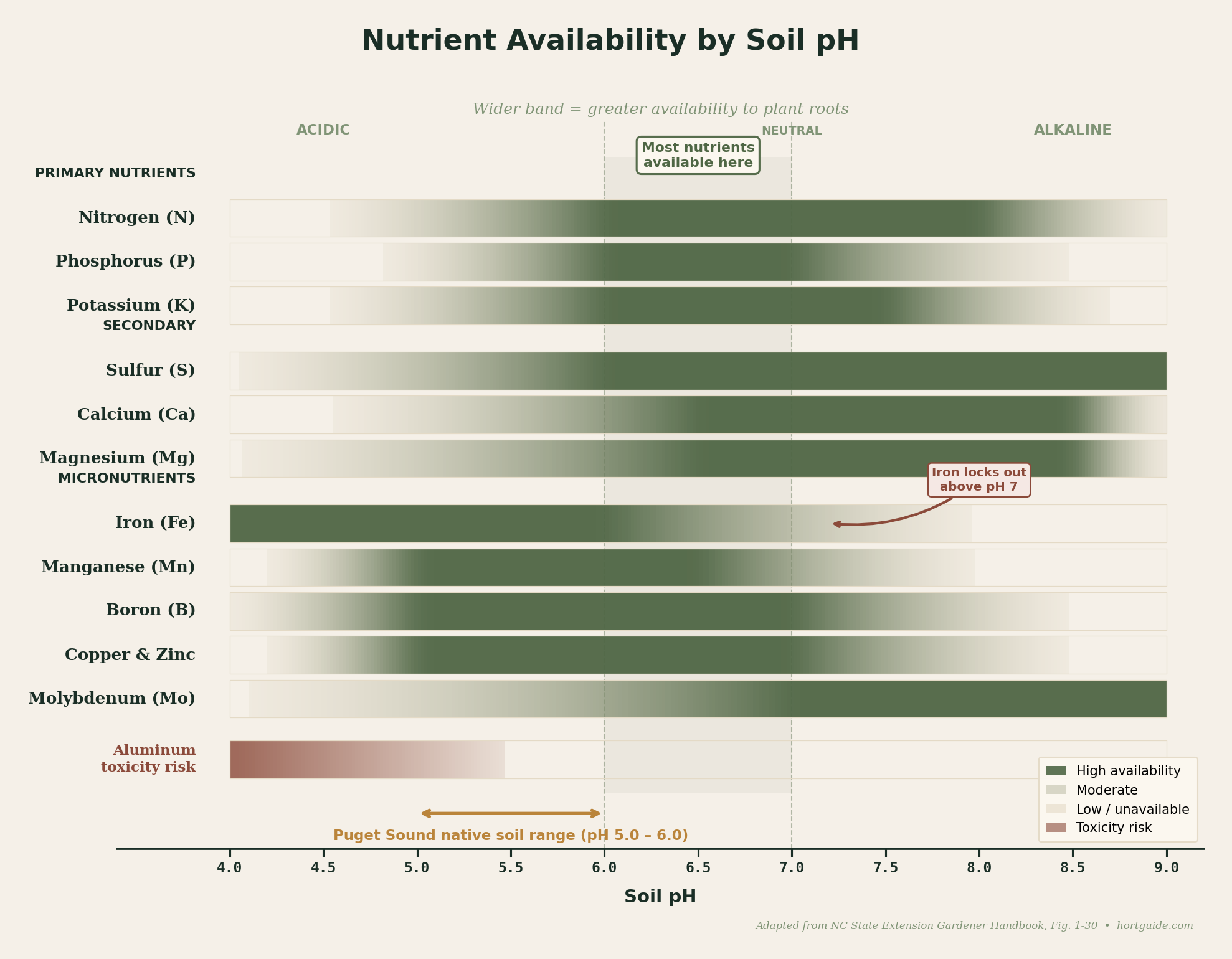 Nutrient availability across the pH spectrum. Most essential nutrients are most accessible between pH 6.0 and 7.0. Iron availability drops sharply above pH 7, which is the mechanism behind interveinal chlorosis on calcifuge species near concrete foundations. Adapted from NC State Extension Gardener Handbook, Figure 1-30.
Nutrient availability across the pH spectrum. Most essential nutrients are most accessible between pH 6.0 and 7.0. Iron availability drops sharply above pH 7, which is the mechanism behind interveinal chlorosis on calcifuge species near concrete foundations. Adapted from NC State Extension Gardener Handbook, Figure 1-30.
There is no single “correct” pH. A blueberry thriving at pH 4.8 is not in bad soil. It is in exactly the right soil for its chemistry. A vegetable garden producing at pH 6.5 is not in better soil. It is in different soil, optimized for a different set of plants. The number only means something when you hold it against what you are trying to grow.
What Your Soil Probably Is
The pattern across this region is consistent: people are surprised their soil is already acidic, and then they are surprised it is not all the same number. The Puget Sound lowlands are not one soil. They are a patchwork left behind by glaciers, and your pH depends on where in that patchwork you garden.
If you are on the upland hills and ridges where most suburban neighborhoods sit, you are almost certainly on Alderwood or Everett. Alderwood, the dominant residential soil from Everett to Tacoma, runs pH 5.8 to 6.0. Most landscape plants do fine at that number without any adjustment. Everett, the gravelly outwash on terraces, is more acidic at 5.3 to 5.6 because its coarse texture lets everything leach faster, including the calcium that would otherwise buffer the acidity. Blueberries planted on Everett soil often need nothing at all.
Down on the valley floors the picture shifts. Woodinville silt loam, the soil everyone in the Green River valley calls “clay” (it is not), runs 5.4 to 5.8. But walk a quarter mile to a river terrace and you hit Puyallup soil at 6.0 to 6.8, the most fertile ground in the region and the only common Puget Sound soil where you might actually need sulfur to grow blueberries. Briscot on floodplains is essentially neutral at 6.6 to 6.8. Two neighbors on the same street can have pH values a full unit apart if one sits on a terrace and the other on a valley floor.
The outlier is Seattle Series organic muck in low-lying bogs and marshes: pH 4.8 to 6.0, the most acidic soil in the region. If you need to lime on Seattle Series soil, prepare to do it every year or two, because the organic material re-acidifies as it decomposes. It is the one soil type where pH management is a permanent commitment rather than a one-time adjustment.
The soil series library has complete profiles for all nine regional series. The soil texture and drainage guide shows you how to identify yours using the USDA Web Soil Survey and three field tests you can do this weekend. Once you know your series, the pH question gets specific.
The soil you inherited vs. the soil you created. Everything above describes native conditions, the pH your soil would have if you left it alone. But nobody leaves soil alone. Concrete foundations leach calcium into adjacent beds and push local pH above 7.0. Foundation beds can test at pH 7.4 while the native soil ten feet away reads 5.6. Years of wood ash dumped in the same garden corner alkalinize a bed that started at 5.5. Over-liming a lawn nudges pH too high for the ornamentals planted ten feet away.
And then there is what I think of as the tap water treadmill. Municipal water in the Puget Sound region typically runs pH 7.5 to 8.5. Every time you irrigate, you push the soil pH slightly upward. On sandy soils with low buffering capacity, this drift is measurable within a few years. On the silt loams and clays it takes longer, but the direction is the same. If you have ever wondered why your hydrangea flowers slowly shifted from blue to pink over the years, this is probably why.
When to Test and How
If you are planting blueberries, rhododendrons, azaleas, heathers, or pieris, test first. Those plants have narrow pH windows and no tolerance for guessing. If you are starting a vegetable garden, test first. Most vegetables perform best between 6.0 and 6.8, and the difference between your soil at 5.4 and the target at 6.2 determines whether you lime and how much. If you are planting landscape trees and shrubs with no particular pH demands, testing is useful but not urgent. Most of what thrives in this region is already adapted to moderately acid soil.
Skip the home pH meter. Spend $25 to $40 on a lab test from WSU Puyallup or the King Conservation District. What you get back is not just a pH number. You get buffer pH, which tells the lab how resistant your soil is to change, and that number determines the amendment rate they recommend. You get organic matter percentage, phosphorus, potassium, and cation exchange capacity. Without the buffer pH, any lime or sulfur rate is a guess. With it, the recommendation is calibrated to your actual soil.
Sampling matters more than people think. Grab soil from six to eight spots across the area you plan to manage, all at six to eight inches deep. Mix them in a clean bucket. Let the composite air dry, bag about a cup, and send it in. If your property has obviously different conditions (raised bed vs. native soil, hilltop vs. low spot, foundation bed vs. open garden), test each zone separately. A single test that averages pH 6.0 across a yard where the foundation bed is 7.2 and the back garden is 5.4 tells you nothing useful about either spot.
Retest every three to five years for established landscapes. Test annually if you are actively amending: blueberry beds, acidified rhododendron plantings, limed vegetable gardens. A $30 soil test is cheap insurance against overshooting in either direction, and overshooting is easier than you think.
Raising pH: When and How to Lime
Most gardeners in this region will never need to lime their ornamental plantings. The situations where liming makes sense are specific:
Vegetable gardens on very acid soil. If your test comes back below 5.8 and you want to grow tomatoes, peppers, brassicas, or legumes, lime moves you into the 6.0 to 6.8 range those crops prefer. This is the most common legitimate reason to lime in the Puget Sound lowlands.
Lawns where moss is winning. Below pH 5.5, turf grasses cannot access enough nutrients to stay dense, and moss fills the gaps. Raising pH to 5.8 or 6.0 gives the grass back its competitive advantage. The early spring lawn care guide covers the full lawn pH and liming program.
Clubroot prevention. Brassica growers dealing with clubroot (Plasmodiophora brassicae) can suppress the pathogen by maintaining soil pH at 6.8 or above. This is a narrow case where deliberate, significant pH elevation is a disease management tool.
Fall is the time. Apply ground limestone in October or November and let our heavy winter rain carry it into the soil profile. By spring planting, the pH shift has had four to five months to develop. Irrigation alone cannot push lime as deep as five months of Pacific Northwest rainfall.
Material choice. Ground agricultural limestone is the standard. Dolomitic lime adds magnesium alongside calcium; choose it only if your soil test shows low magnesium. Calcitic lime adds calcium only. Both work at the same rate. Wood ash raises pH but its calcium content varies wildly from one fire to the next, and it is the single easiest amendment to over-apply. I have seen beds pushed above pH 7.5 from years of casual ash dumping. If you use wood ash at all, test first and go light.
Rates and the buffer capacity problem. OSU Extension recommends 5 to 10 pounds of ground limestone per 100 square feet as a starting rate. Never exceed 50 pounds per 1,000 square feet in a single application. Heavier doses create a pH spike at the surface while leaving the root zone unchanged six inches down.
Here is where your soil series matters again. Bellingham clay has the highest buffer capacity of any regional soil: high CEC, fine particles, strong resistance to change. A single lime application barely moves the needle. Lighter, more frequent applications (5 pounds per 100 square feet, repeated annually) work better than dumping the whole bag once. On Everett gravelly sandy loam, the opposite: low CEC, low buffer capacity, the pH moves quickly, but the effect fades just as quickly as lime leaches through the coarse texture. Sandy soils need smaller doses repeated more often. Clay soils need patience and repetition. The lab’s buffer pH measurement is what tells you which pattern your soil follows.
Retest before adding more. Response time is three to six months, longer on heavy soil, shorter on sand. Adding a second application before the first has taken full effect is the most common over-liming mistake, and it is harder to fix than under-liming.
Lowering pH: When and How to Acidify
Three situations call for acidification here:
Planting blueberries or ericaceous shrubs. Target pH 4.5 to 5.5. On Alderwood or Everett soil, you may already be there. On Puyallup or Briscot soil, you are not, and sulfur is necessary. The blueberry selection guide covers species-specific amendment, soil scenarios, and cultivar selection in detail; what follows here is the general framework that applies to any acid-loving planting.
Beds next to concrete foundations. If a foundation bed tests at pH 7.2 when the surrounding soil is 5.8, calcium leaching from the concrete is the source. The yellow-veined rhododendron leaves you see in these beds are iron chlorosis: the plant’s signal that pH has locked out the micronutrients it needs. Acidifying the bed, not fertilizing, is the fix.
Sites with imported construction fill. New-construction soils are some of the most pH-disrupted sites I work on. Imported fill runs alkaline more often than not. If your test on a new-construction lot returns pH 7.0 or above while the native soils around it are in the 5s, the fill is the problem, and the trees and shrubs planted into it will show the stress within a few years.
Elemental sulfur is the standard amendment. It is inexpensive and effective, but it works on biology’s schedule, not yours. Soil bacteria (Thiobacillus species) must oxidize the sulfur into sulfuric acid before it changes pH, and that microbial process requires warmth and oxygen. In this region, that means spring application (March through April) is more effective than fall. Our cold, wet winters slow soil biology nearly to a halt; sulfur applied in October may sit unchanged until the soil warms in March. This is the opposite of lime timing: lime in fall to ride the rain, sulfur in spring to ride the biology.
Rates depend on your soil texture. To lower pH by half a unit on sandy soil, apply about 0.5 pounds of elemental sulfur per 100 square feet. On silt loam, double that. On clay, triple it. The difference is buffer capacity again: fine-textured soils resist change, so they need more amendment to move the same distance. Apply no more than 2 pounds per 100 square feet at a time and retest after three to six months before adding more. Patience matters here. Sulfur overdose drops pH below the target and creates the aluminum toxicity problem you were trying to avoid.
Iron sulfate works faster because it does not need microbial conversion. Use it for small, targeted adjustments: a single foundation bed, not a whole property. It costs more per unit of pH change and is easier to over-apply.
Ammonium sulfate is the maintenance tool. It supplies nitrogen and slowly acidifies as the ammonium converts to nitrate, about 0.2 to 0.3 pH units per season at normal fertilizer rates. That is not enough to make a large correction, but it is enough to counteract the upward drift from tap water irrigation. The blueberry selection guide covers how this fits into a full acidification program for ericaceous plantings.
The coffee grounds myth. It shows up in every gardening forum: save your coffee grounds, sprinkle them around your acid-loving plants, and the pH will drop. Used coffee grounds are nearly neutral (pH 6.5 to 6.8). They have almost no acidifying effect. The myth confuses the acidity of brewed coffee with what remains in the spent grounds after the acids have been extracted into your cup. Coffee grounds are fine as mulch or compost feedstock. They are not a pH amendment. Vinegar is worse: it acidifies momentarily, kills soil biology, and the effect disappears within days. Aluminum sulfate is widely sold but can reach toxic aluminum concentrations at the rates needed for meaningful pH change. Stick with elemental sulfur for initial adjustment and ammonium sulfate for maintenance.
Keeping pH Stable Without Constant Amending
If you find yourself liming or acidifying every year, something upstream is working against you. The goal is a soil that holds its own pH through its organic matter and microbial community, not through annual chemical intervention.
Organic matter is the long game. Compost, mulch, and decomposing wood chips build the soil’s cation exchange capacity, its ability to hold nutrients and resist pH swings in either direction. A soil with 5 percent organic matter resists drift from tap water, concrete leaching, and over-amendment far better than a soil with 2 percent. This is why the practices covered in the mulching guide and the soil texture guide keep showing up as the foundation of every soil management strategy: building organic matter is the slow, permanent version of what lime and sulfur do temporarily.
Match fertilizer to your pH goal. Ammonium-based fertilizers acidify slightly with every application. Nitrate-based fertilizers push pH slightly upward. If you are maintaining an acidified bed, choose the form that reinforces the direction you want rather than working against it.
Healthy soil biology compensates for imperfect pH. The mycorrhizal partnerships that help roots access nutrients work best in undisturbed, high-organic soil, the same conditions that stabilize pH. Building soil biology and building pH stability are not separate projects. They are the same project.
When to stop chasing a number. If your plants look healthy, your pH is fine regardless of what the test says. A rhododendron with deep green leaves and vigorous growth at pH 5.8 does not need acidification to hit a textbook-ideal 5.0. A vegetable garden producing well at pH 5.9 does not need lime to reach 6.5. The soil test guides your decisions. It does not replace what you can see with your own eyes.
| Timing | Action | Notes |
|---|---|---|
| Late winter (Feb) | Send soil samples to lab | Results back before spring planting decisions |
| Early spring (Mar-Apr) | Apply elemental sulfur if acidification needed | Soil biology activating; warm enough for microbial oxidation |
| Spring planting | Match plant pH preferences to tested beds | Use soil series library and plant profile pH fields |
| Fall (Oct-Nov) | Apply lime if soil test indicates need | Winter rain carries lime into the profile |
| Every 3-5 years | Retest established beds | Annually if actively amending |
Sources
- Why soil pH matters, and how to manage it. Oregon State Extension.
- Soils & Plant Nutrients (Extension Gardener Handbook, Ch 1). NC State Extension. Nutrient availability diagram, lime rate guidance.
- pH Problems: Acid and Alkaline Soil. UC IPM.
- Soil Acidification: How to Lower Soil pH (AGF-507). Ohio State Extension. Sulfur rate table by soil type.
- Calculating the Rate of Acidifiers. NC State Extension.
- Soil Acidity in the PNW. Sky Nursery, Shoreline, WA.
- Chalker-Scott, L. A Gardener’s Primer to Mycorrhizae (FS269E). WSU Extension, 2017.
- USDA-NRCS Official Series Descriptions: Alderwood, Woodinville, Everett, Puyallup, Bellingham, Seattle, Briscot, Norma, Snohomish.
Always read product labels before applying any soil amendment. WSU Extension and your local conservation district offer soil testing and amendment guidance specific to your site.
